SHANGHAI, China, February 27, 2026 — Asieris Pharmaceuticals (Stock Code: 688176.SH), a global innovative pharmaceutical company specializing in genitourinary tumors and women’s health, announced today that its marketing authorization application (MAA) for APL-1702 (CEVIRA) for the treatment of high-grade squamous intraepithelial lesions (HSIL) has been accepted by the European Medicines Agency (EMA).

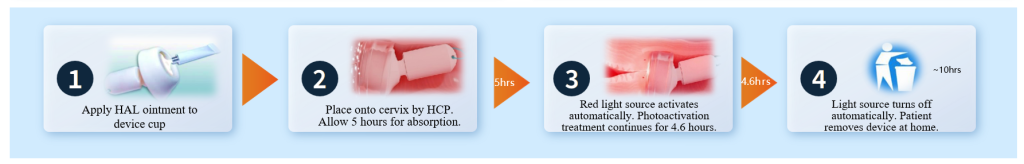
APL-1702 is a globally first-in-class, China-originated photodynamic therapy(PDT) product. As a drug-device co-packaging product, it integrates hexaminolevulinate hydrochloride ointment with a disposable, single-use cervical photodynamic light applicator. Through its innovative localized drug delivery and intravaginally positioned cold light source, APL-1702 represents a breakthrough in the treatment paradigm of HSIL..
Designed for convenient “treat-and-go” outpatient use, the therapy is administered by gynecologists during a routine outpatient visit without the need for anesthesia, the entire placement procedure is typically completed within 10 minutes. After device placement, patients may immediately return to normal work and daily activities and are not required to remain in the hospital during the illumination period. Upon completion of treatment, the device can be safely removed by the patient at home.
This “short outpatient placement followed by at-home treatment completion” model substantially improves clinical efficiency and treatment accessibility, and supports broader implementation in primary and community healthcare settings.
Additionally, APL-1702 incorporates an innovative cold light source technology that enhances treatment comfort by maintaining tissue temperature at the treatment site below 42°C, Clinical data indicate that 97% of patients reported no pain in the cervical treatment area, with no observed cervical structural damage, thereby avoiding the risk of thermal injury associated with conventional photodynamic therapy, minimizing local irritation, and significantly improving overall patient tolerability.
The MAA submitted to the EMA is primarily supported by data from an international, multicenter Phase III clinical trial of APL-1702, which included more than 20% patients from Europe.
The pivotal study results were formally published in December 2025 in Med, a Cell Press journal. The study was led by Academicians Lang Jinghe and Zhu Lan from Peking Union Medical College Hospital, with Professor Peter Hillemanns from Hannover Medical School serving as a senior author.
In addition, the clinical data were disseminated through invited oral presentations at several major international conferences, including the European Research Organization on Genital Infection and Neoplasia (EUROGIN) 2024, the Society of Gynecologic Oncology (SGO) Annual Meeting 2024, and the Photodynamic Therapy & Photodiagnosis Update 2024.
Further academic exchanges and scientific discussion with basic researchers and clinical specialists from China and abroad took place at the 27th National Clinical Oncology Conference & 2024 CSCO Annual Meeting, the International Papillomavirus Conference (IPVC) 2024, and the 37th Public Health Symposium 2025.
“The acceptance of APL-1702’s European marketing application in Europe further underscores Asieris’ strong and enduring commitment to addressing critical unmet medical needs on a global scale,” said Joanna Zhang, Chief Medical Officer of Asieris Pharmaceuticals. “Building on our deep and proven expertise in the development of drug-device combination products, we have successfully advanced a truly non-invasive, patient-centred therapy from clinical research to the European regulatory review stage. This important milestone validates both our strategic focus and our ability to execute with discipline and rigor.. We remain firmly committed to accelerating the global development of APL-1702, with the goal of bring this transformative treatment option to women worldwide and contributing meaningfully to global efforts toward cervical cancer elimination.”
About Asieris
Asieris Pharmaceuticals(688176.SH), founded in March 2010, is a global biopharma company specializing in discovering, developing and commercializing innovative drugs for the treatment of genitourinary tumors and women’s health. We strive to improve human health to preserve patient’s dignity. We aim to become a global pharma leader that integrates R&D, manufacturing and commercialization in our areas of focus.
Through proprietary R&D and strategic collaborations, the company is committed to developing first-in-class drugs and other innovative technologies and products that address significant unmet medical needs, building a robust portfolio spanning from disease diagnosis to treatment within its focused therapeutic areas. Driven by patient needs, we are comprehensively advancing Commercialization 2.0 to improve the accessibility and affordability of innovative products, thereby benefiting more patients in China and globally.